Fuels
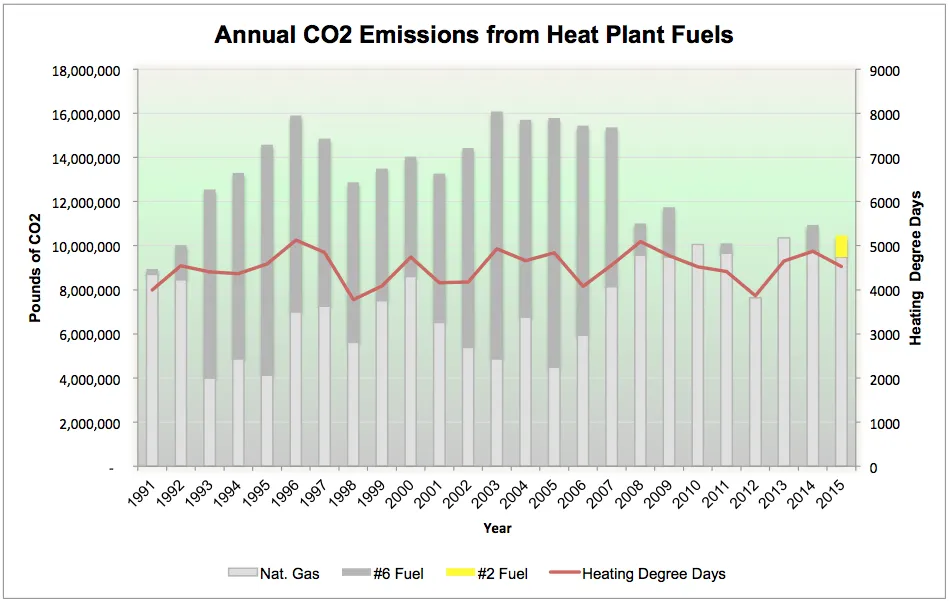
Comparing the two fuels that we have burned over the last two decades it is clear to see that burning oil raises the carbon profile of the College while burning natural gas lowers it. You can also download a larger PDF version of the above chart.
Another way of looking at fuels is to consider the carbon content. All fuels contain carbon bound to hydrogen atoms. Oxidizing those compounds releases the carbon which is commonly referred to as CO2. More properly they are referred to as carbon equivalents to cover the broad spectrum of greenhouse gases that are created when fuel is burned. It's important to know that fuels for a given Btu content have different stack characteristics. Processed natural gas is a particularly clean burning fuel. It has very little sulfur, no ash and not much comes out of the stack but carbon dioxide, carbon monoxide and water vapor. Natural gas actually has no scent. The "rotten egg" smell that we all associate with natural gas is the addition of T-butyl Mercaptan which became a standard practice after the 1937 disaster at the New London School in Texas.
The college made the decision in 2014 to eliminate #6 heavy fuel oil as our alternate fuel. One look (and sniff) of a jar full of #6 will demonstrate it is a much different fuel. It is considered to be one step above the lowest grade of fuel oil called bunker C, commonly used in the shipping industry to fuel the massive engines found in ocean faring ships. In place of the #6 fuel the college has converted to #2 fuel oil. It has a fraction of the sulfur and ash content of the heavy oil so offers a much cleaner burn with far less stack emissions. Our primary fuel will remain natural gas for the foreseeble future but it is a step forward that #6 has joined coal as a legacy fuel in our history.



