Kathy Siwicki
Howard A. Schneiderman '48 Professor Emerita in Biology
Emeritus Biology
Contact

Courses | Research | Major Publications
Education
ScB Biology, Brown University
MPhil Pharmacology, Cambridge University
PhD Neurobiology, Harvard University
![]() Courses
Courses
Biology 1: Cellular and Molecular Biology
BIOL 022. Neurobiology. A study of the fundamentals of modern neuroscience, from the molecular to the systems levels. The electrical and chemical signaling properties of neurons and their underlying cellular and molecular mechanisms are emphasized in the first half of the semester, and the functional organization of selected neural systems are considered in the second half. Weekly laboratories involve neuroanatomical and neurophysiological techniques, as applied to invertebrate nervous systems.
BIOL 122. Developmental Neurobiology. Cellular and molecular mechanisms of nervous system development are the focus of this seminar, involving extensive reading and discussion of the research literature and independent laboratory projects.
BIOL 123. Learning and Memory. The neural systems and cellular processes involved in different types of learning and memory are studied through extensive reading and discussion of the research literature. Students also develop research proposals and work in teams to conduct independent research projects.
BIOL 001. Cellular and Molecular Biology. A team-taught introduction to the study of living systems at the cellular and molecular level. The fundamentals of biochemistry, cell biology, molecular biology, and genetics are illustrated in both lectures and labs with examples drawn from microbiology, neurobiology, and developmental biology.
![]() Research
Research
Neural Mechanisms of Courtship and Memory in Drosophila
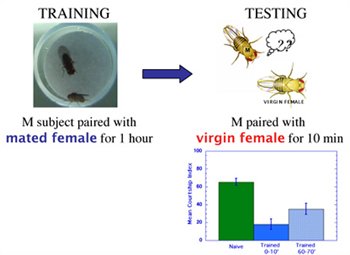
Research in the Siwicki lab focuses on understanding the neurobiology of courtship behavior and memory in the fruit fly, Drosophila melanogaster. The behavioral responses of wild-type male fruit flies to the aphrodisiac properties of female flies are largely instinctive - males reared in social isolation actively court virgin females. But male flies often encounter sexually attractive individuals that are not receptive to mating, e.g., immature virgin females, or mature females that recently mated with another male. In fact, males can learn from their attempts to court unreceptive targets, and the neural processes responsible for this learning are the focus of our interests. Specifically, we aim to identify the neural circuits and mechanisms of synaptic plasticity responsible for a form of associative learning known as courtship conditioning. In this learning paradigm, after males are paired with an unreceptive, previously mated female for an hour, they suppress their courtship of other females for 1-2 hours (Seigel and Hall, 1979).
Several years ago we evaluated the role of the mushroom bodies (MB) in courtship conditioning by pharmacologically ablating the MBs in early larval development. Our results indicated that the MBs are not required for innate male courtship behavior, but are required for normal memory of courtship conditioning (McBride et al. 1999). MB-ablated males showed only a transient memory
of conditioning; their courtship behavior returned to naïve levels within 30 minutes after training. Similar results were obtained in more recent studies in our lab, involving GAL4/UAS manipulations of MB function. The evidence suggests that memory assayed immediately after training (0 minutes) is independent of MB function, while memory lasting 30 minutes or longer requires MB processing.
We also described two novel training protocols that produced long-term memory of courtship conditioning (McBride et al., 1999). One consists of a continuous 5-h pairing with a mated female. The other consists of 3 sets of 1-h pairings, interrupted by 30-60 min breaks. For up to 8 days after these intensive training regimes, males showed a significant suppression of courtship
activity relative to age-matched naïve controls. Other researchers have adopted our training procedures to assess the roles of other genes in long-term memory (Presente et al., 2004; Sakai et al., 2004). Meanwhile, students in our lab continue to investigate additional training paradigms for inducing both short-term and long-term courtship memory.
In spite of the likely ecological relevance of courtship conditioning, progress in deciphering its neural mechanisms has been impeded by its complexity, specifically by the multisensory nature of the training experience with mated females. We have begun to define the relevant sensory cues in collaboration with Jean-Francois Ferveur and coworkers at the Universite de Bourgogne in Dijon, France. Using flies of different genotypes that express distinct cuticular hydrocarbon blends (Ferveur et al., 1997), we have identified a specific role for female cuticular pheromones as a conditioned stimulus in courtship conditioning. With current molecular and genetic tools for identifying the sensory neural systems that transduce sex pheromones (Bray and Amrein, 2003), these studies will contribute to understanding the behavioral significance of different components of the pheromonal blend in innate vs. learned aspects of courtship behavior.
Literature CitedBray, S. and Amrein, H. 2003. A putative Drosophila pheromone receptor expressed in male-specific taste neurons is required for efficient courtship. Neuron 39: 1019-1029.
McBride, S.M.J., Giuliani, G., Choi, C., Krause, P., Correale, D., Watson, K., Baker, G. and Siwicki, K.K. 1999. Mushroom body ablation impairs short-term memory and long-term memory of courtship conditioning in Drosophila melanogaster Neuron 24: 967-977.
Presente, A., R.S. Boyles, C.N. Serway, S.J. deBelle, A.J. Andres, 2004, Notch is required for long-term memory in Drosophila, Proc. Natl. Acad. Sci. USA 101:1764-1768
Sakai, T., T. Tamura, T. Kitamoto, and Y. Kidokoro, 2004, A clock gene, period, plays a key role in long-term memory formation in Drosophila. Proc. Natl. Acad. Sci. USA 101: 16058 -16063
Siegel, R.W. and Hall, J.C. 1979. Conditioned responses in courtship behavior of normal and mutant Drosophila. Proc. Natl. Acad. Sci. USA 76: 3430-3434.
Tompkins, L., Siegel, R.W., Gailey, D.A. and Hall, J.C. 1983. Conditioned courtship in Drosophila and its mediation by association of chemical cues. Behav. Genet. 13: 565-578.
![]() Major Publications
Major Publications
(* indicates undergraduate co-authors)
Siwicki, K.K. and C.A. Bishop, 1986, Mapping of proctolin-like immunoreactivity in the nervous systems of lobster and crayfish. J. Comp. Neurol. 243: 435-453
Marder, E., S.L. Hooper, and K.K. Siwicki, 1986, Modulatory action and distribution of the neuropeptide proctolin in the Crustacean stomatogastric nervous system. J. Comp. Neurol. 243: 454-467
Siwicki, K.K., B.S. Beltz, and E.A. Kravitz, 1987, Proctolin in identified serotonergic, dopaminergic, and cholinergic neurons in the lobster, Homarus americanus. J. Neurosci. 7: 522-532
Siwicki, K.K., C. Eastman*, G. Petersen, M. Rosbash, and J.C. Hall, 1988, Antibodies to the period gene product of Drosophila reveal diverse tissue distribution and rhythmic changes in the visual system. Neuron 1: 141-150
Siwicki, K.K., S. Strack, M. Rosbash, J.C. Hall, and J.W. Jacklet, 1989, An antibody to the Drosophila period protein recognizes circadian pacemaker neurons in Aplysia and Bulla. Neuron 3:51-58
Zerr, D.M.*, M. Rosbash, J.C. Hall, and K.K. Siwicki, 1990, Circadian fluctuations of period protein immunoreactivity in the CNS and visual system of Drosophila. J. Neurosci. 10: 2749-2762
Siwicki, K.K., W.J. Schwartz, and J.C. Hall, 1992, An antibody to the Drosophila period protein labels antigens in the suprachiasmatic nucleus of the rat. J. Neurogenet. 8: 33-42
Rosewell, K.L., K.K. Siwicki, and P.M. Wise, 1994, A period (per)-like protein exhibits daily rhythmicity in the suprachiasmatic nuclei of the rat. Brain Research 659: 231-236
Hardin, P. and K.K. Siwicki, 1995, The multiple roles of per in the Drosophila circadian clock, Seminars in the Neurosciences, Vol. 7(1), pp. 15-25
Emery, I.F., J.M. Noveral, C.F. Jamison, and K.K. Siwicki, 1997, Rhythms of Drosophila period gene expression in culture, Proc. Natl. Acad. Sci. USA 94: 4092-4096
McBride, S.M.J.*, G. Giuliani*, C. Choi, P. Krause*, D. Correale*, K. Watson*, G. Baker* and K.K. Siwicki, 1999, Mushroom body ablation impairs short-term and long-term memory of courtship conditioning in Drosophila. Neuron 24: 967-977
Wise, S.*, N.T. Davis, E. Tyndale*, J. Noveral, M.G. Folwell, V. Bedian, I.F. Emery and K.K. Siwicki, 2002, Neuroanatomical studies of period gene expression in the hawkmoth, Manduca sexta. J. Comp. Neurol. 447:366-380.
Dockendorff, T.C., H.S. Su, S.M.J. McBride, Z. Yang, C. Choi, K.K. Siwicki, A. Sehgal, and T.A. Jongens, 2002, Drosophila lacking dfmr1 activity show defects in circadian output and fail tomaintain courtship interest. Neuron 34: 973-984
Siwicki, K.K. and L. Ladewski*, 2003, Associative learning and memory in Drosophila: Beyond olfactory conditioning, Behavioural Processes 64: 225-238
Mc Bride, S.M.J., C.H. Choi, Y. Wang, D. Liebelt, E. Braunstein, D. Ferrero, A. Sehgal, K.K. Siwicki, T.C. Dockendorff, H.T. Nguyen, T.V. McDonald and T.A. Jongens, 2005, Pharmacological rescue of synaptic plasticity, courtship behavior and mushroom body defects in a Drosophila model of Fragile X Syndrome. Neuron 45: 753-764
Siwicki, K.K., P. Riccio*, L. Ladewski*, F. Marcillac, L. Dartevelle, S.A.
Cross* and J.-F. Ferveur, 2005, The role of cuticular pheromones in
courtship conditioning of Drosophila males, Learning & Memory 12: 636-645